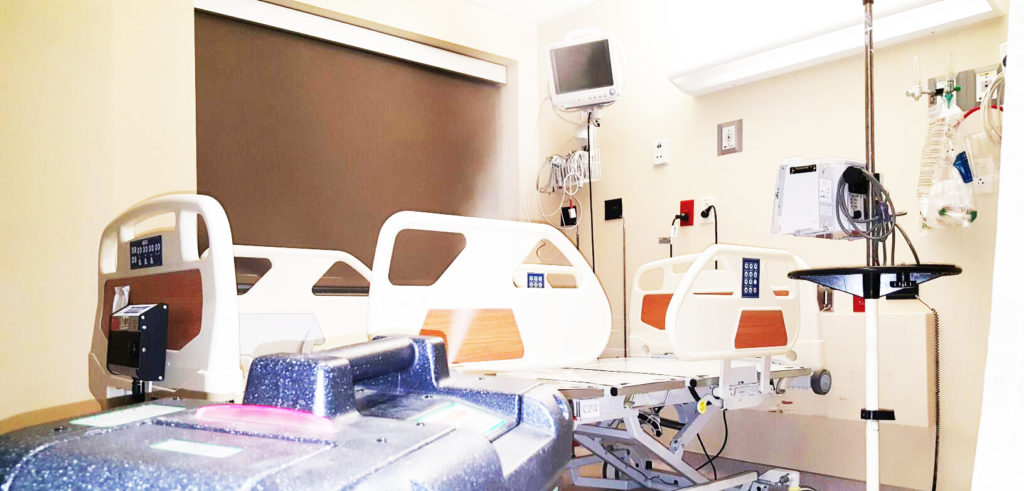
Decontamination company CURIS® System is working to find innovative ways to help in the fight against the virus.

As hospitals prepare for the coming coronavirus surge, personal protective equipment (PPE) is in high demand; in particular N95 respirators and surgical masks, which are used to protect the wearer from airborne particles and from liquid contaminating the face. In a recent survey by Premier Inc., a shortage in N95 masks was the top concern among hospitals and health systems treating COVID-19 patients. Premier calculated a potential surge need of up to 17X for these masks, noting that the availability of PPE and burn rates for PPE products were the two most commonly cited “surprises” of the pandemic.
That’s why the ability to re-sterilize equipment is critically important to supporting health systems through the crisis – and just one way that decontamination company CURIS® System is working to find innovative ways to help in the fight against the virus.
CURIS® System is enabling the re-sterilization of N95 masks, allowing hospitals to quickly and efficiently eliminate harmful pathogens from thousands of masks at a time – a game changer in helping hospitals be adequately supplied during the pandemic. Under normal circumstances, N95 masks are discarded after each use; however, re-sterilizing this equipment extends its usage considerably, increasing the available supply for patients.
CURIS® partnered with SPIRE to build sealed chambers that contain the masks while the company’s patented product and delivery system sterilizes them. The system acts as a fog, coating the entirely of the space or enclosure being decontaminated. This product can re-sterilize over 1,000 masks at a time and over 4,000 per day. The product is currently being fast-tracked for FDA approval through the Emergency Use Authorization due to the immense need from hospitals for this PPE to handle the coming coronavirus surge.

The company’s facility has been fully functional and operating around the clock for the last six days and seven days a week for the last three weeks, even working on Sundays for free to assist local nonprofits in decontamination efforts.
We understand that the current circumstances have placed a large amount of responsibility on companies like ours, and we are grateful for the opportunity to be of assistance during this global crisis.
Frances Grinstead, Founder and CEO of CURIS® System
As businesses begin to plan for safe re-opening of buildings and facilities, decontamination education is critically important in order for customers to feel safe. The company has seen an increase in interest and opportunity for using its products to ensure the safe return of customers to large gathering spaces. One of the company’s clients is a Hard Rock Café in Oklahoma, which regularly sterilizes its entire property with the CURIS® System.
“it’s amazing to see the ingenuity of some of our clients in finding creative ways to tackle the challenges of a crisis like COVID-19,” said Grinstead. “We’re learning new ways that our technology has been applied every day.”
CURIS® System manufactures its decontamination equipment and solution in Oviedo. The company has products used in hospitals around the country, and its aerosol product is the only EPA-certified method to kill 99.9999% of super bug C. diff in a tri-part soil load.